Apple Classroom got an exciting update this week: Agendas! Agendas allow a teacher to create a workflow for students. Here's a look at how they work.
When you log into Apple Classroom and start a class, you see a view like the image below.

Under the Classes listing on the left, you will now see Agendas. Click on the word Agendas to access or create an agenda. You will see the agendas you have created or click the Add Agenda button to create a new one. In my classroom, Review Day always takes the same format, so I created an agenda called Review Day.
After creating the agenda, you can customize it by adding time limits and alerts, send students to websites, open certain apps, and lock or unlock devices. You can even create agendas for particular groups of students. Here are images that show of some of the options and the beginnings of my Review Day Agenda.
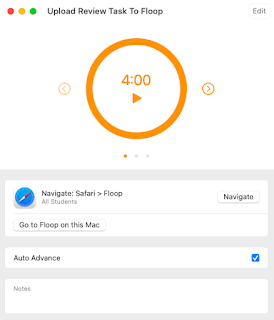
If you look at the My Agenda image, you will see that I have assigned 4 minutes for students to navigate to Floop and upload an image of their Review Task. After 4 minutes, the agenda will Auto Advance to the next item which is a pause for directions. During this 1 minute pause, the students devices will lock so I can try to gain their full attention. After directions, students will return to Floop to participate in a peer review of the Review Tasks. Again, this step is timed. When the time runs out, I can receive an alert or not, depending on how I create the agenda.
Now that the agenda has been created, it's time to use it in a class. When I am logged into Apple Classroom, I start a class. Once inside the class, I can click the people icon (top left corner) to see my students' screens or I can click the new agendas icon (just to the right of the people) to see agendas I have created.
When I click the agenda icon, I see this screen:
I click the Open Agenda button and select my Review Day Agenda. The agenda opens and I get a play button so I can begin the process in class. Notice the small arrows next to the 4:00 timer. These allow me to skip a step in my agenda if needed.
Agendas has great potential for managing classroom routines. Agendas are a great way to pace a lesson, especially on days with lots to do. Sometimes I mean to use a timer or limit discussion to a certain amount of time. I love that I can plan that out ahead of time and let the Agenda serve as my reminder. I think they'd be especially great for station rotations or centers work because they would allow a teacher to have an automated teacher's assistant. I also like the idea of creating workflows for days that have the same steps like my Review Day. Since Apple Classroom can be joined by students who are absent, Agendas would also be a great way to help a student know what's happening at each stage of class despite being in a different location. I'm looking forward to trying these out in my classroom next fall. What agendas will you create? Use the comments to share your ideas.